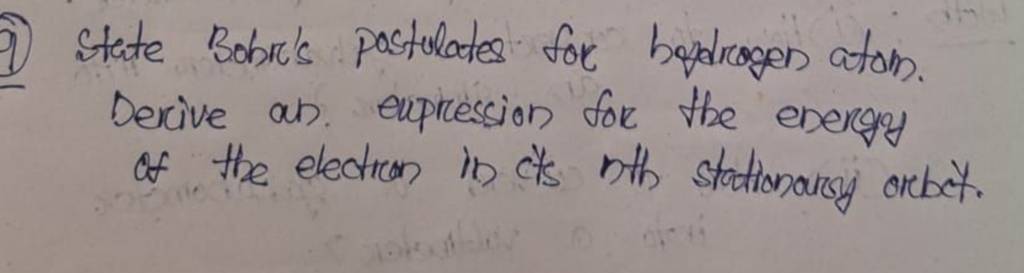 _0.jpg)
The Bohr model of the atom, a radical departure from earlier, classical descriptions, was the first that incorporated quantum theory and was the predecessor of wholly quantum-mechanical models. 004 - The Bohr AtomIn this video Paul Andersen describes the major parts of an atom and explains how the Bohr Model more accurately represents the location o. The existence of the atomic spectra is support for Bohrs model of the atom. It is analogous to the structure of the Solar System, but with attraction provided by electrostatic force rather than gravity, and with the. After the discovery of fission, Bohr was acutely aware of the theoretical possibility of making an atomic bomb. In this expression, \(k\) is a constant comprising fundamental constants such as the electron mass and charge and Planck’s constant. Bohr model, description of the structure of atoms proposed in 1913 by the Danish physicist Niels Bohr. Bohrs model suggests that the atomic spectra of atoms is produced by electrons gaining energy from some source, jumping up to a higher energy level, then immediately dropping back to a lower energy level and emitting the energy difference between the two energy levels. In atomic physics, the Bohr model or RutherfordBohr model of the atom, presented by Niels Bohr and Ernest Rutherford in 1913, consists of a small, dense nucleus surrounded by orbiting electrons. The motion of the electrons in the Rutherford model was unstable because. Bohr’s expression for the quantized energies is: In 1913, just two years after the Rutherford atomic model had been introduced, Danish physicist Niels Bohr, a student of Rutherford’s, proposed his quantized shell model of the atom (see Bohr model) to explain how electrons can have stable orbits around the nucleus. Instead of allowing for continuous values for the angular momentum, energy, and orbit radius, Bohr assumed that only discrete values for these could occur (actually, quantizing any one of these would imply that the other two are also quantized). Learn how the Bohr model of the atom explains the emission and absorption of certain frequencies of light from the elements using photon frequency calculations. The absolute value of the energy difference is used, since frequencies and wavelengths are always positive. In this equation, h is Planck’s constant and E i and E f are the initial and final orbital energies, respectively.  
In 1913, after returning to Copenhagen, he began publishing his theory of the simplest atom. Bohr, shown in Figure 22.8, became convinced of its validity and spent part of 1912 at Rutherford’s laboratory. When the electron is in this lowest energy orbit, the atom is said to be in its ground electronic state (or simply ground state).\] Niels Bohr (18851962), a Danish physicist, did just that, by making immediate use of Rutherford’s planetary model of the atom. Thus, the electron in a hydrogen atom usually moves in the n = 1 orbit, the orbit in which it has the lowest energy. One of the fundamental laws of physics is that matter is most stable with the lowest possible energy. Bohr’s model of the atom accounted for the general chemical properties of the elements, even leading to the discovery of a new elementhafnium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
